Implementation
Work plan – Work package and deliverable
MEDICOR strategically started its BABYLIFE Plus project 3 years ago, starting with a detailed market and need analysis and the definition of the roadmap for the development and market introduction of this new intensive care product. In the past two years the focus was on technology development and proof of concept demonstration.
The work plan presented here is based on the upcoming phase of our strategic roadmap, planned to start after the August milestone of our internal work plan, which is the completion of all testing with the current beta prototype.
The EC founding enables us to speed up this final phase by extending available R&D resources. For these two years’ work plan, we plan open additional job positions already now, which were originally planned for the production and commercialization phase, thus the new jobs will be maintained in long term.
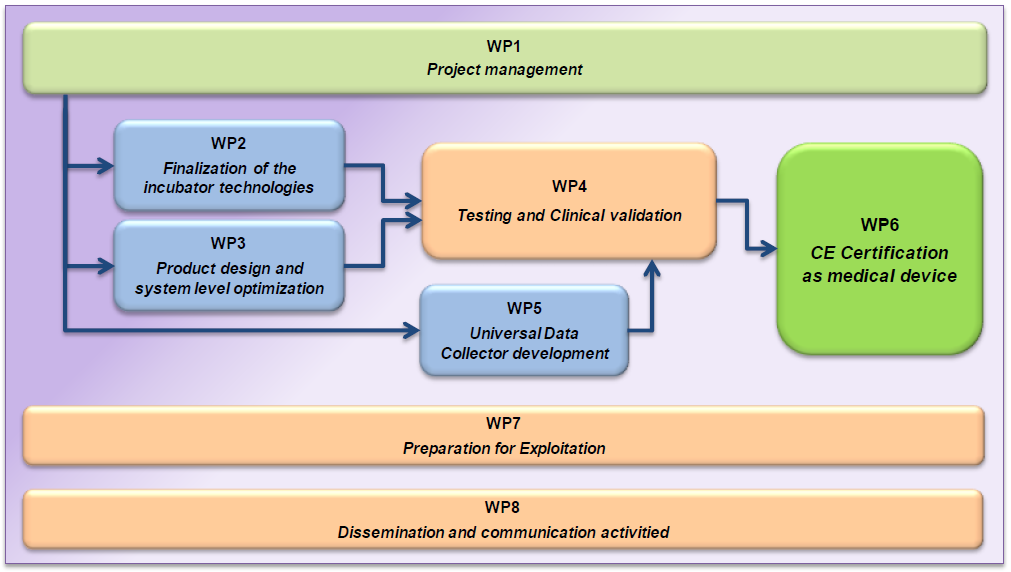
WP1 consolidates all project management tasks, including the coordination with EC as well as technical and administrative project management and contract administration tasks. WP2 and WP3 focus on the final product development upgrading the current Beta prototype to a null-series prototype. This is necessary because all certification tests and also the clinical validation need to be done with prototypes equivalent to the planned final product. Otherwise the process would need to be repeated at least partly with the final design. Thus the main aim of WP2 and WP3 is to finish up the product development already considering future manufacturing strategy and to produce 10 prototypes for testing and clinical validation in WP4 and for dissemination and demonstration in WP8.
The main objective of this project is to complete the required testing, certification and clinical validation tasks. These tasks are focused in WP4. A careful step by step testing approach is necessary in order to ensure maximal infant safety during the clinical studies.
We will start with very detailed quality and performance specification testing according to our quality management process, which will result in justified self-certification of the claimed incubator features and performance parameters. In connection to this we will also manage all legally required testing executed by certified service providers, as well as conformity tests required by the desired CE markings. These tests are mandatory to be completed according to the regulations before the new incubator enters clinical validation.
An important part of WP4 will be the clinical approbation and the extended clinical study. The clinical validation tasks will be executed at SOTE which is the largest university clinic in Hungary having very good international reputation also providing international education programs for the increasing number of European students. The clinical approbation is basically the operation of the incubator in clinical setting, but with a dummy patient. Once the approbation is completed, we will start the actual clinical validation studies with real patients.
WP5 aims the development of a Universal Data Collector until. This was objective added based on the feedback obtained during our recent market survey, which formulated the need for unified patient oriented and systematic data collection. We will finish up the already existing prototype and add the necessary instrument protocols to the software with the aim to utilize these units already during the clinical validation tasks.
The last 3 WPs focus on the implementation of the first key steps of our go-to-market strategy. Namely, preparing and initiating the CE marking process, as well as exploitation and dissemination related activities. Proper innovation management gets important focus though out the project, as well as fine adjustments to our business plan and marketing strategy. We will prepare the road for market entry by intense pre-marketing and dissemination activates. WP8 also includes general communication activities, to ensure the public is informed about this EU founded project.